Washington: Earlier this week, 55 Members of Congress signed on to a letter that calls on US Food and Drug Administration (FDA) Commissioner Margaret Hamburg, M.D. to require the labeling of genetically engineered (GE) food.
The bicameral letter, led by Senator Barbara Boxer (D-CA) and Representative Peter DeFazio (D-OR), was written in support of a legal petition filed by the Center for Food Safety (CFS) on behalf of the Just Label It campaign and its nearly 400 partner organizations and businesses, including Beyond Pesticides. So far, over 950,000 people have submitted comments in support of labeling since CFS filed the legal petition in October 2011.
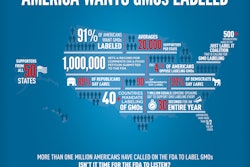
The campaign also recently launched a new infographic, which visually explains why FDA should "Just Label It". It is designed to clearly show the need for labeling of GE foods, and is convenient for sharing on-line and via social media.
Meanwhile, on the West coast, The California Right to Know Initiative Campaign is also gaining momentum with its signature gathering efforts across the state to place a voter backed initiative on the November 2012 ballot that would mandate labeling of genetically engineered food. The effort is supported by a broad coalition of public health, family, environmental organizations, farmers and individuals. So far, thousands of energized volunteers have been collecting signatures to qualify the petition for the ballot since mid-February across the state.
The Congressional letter to FDA states:
"At issue is the fundamental right consumers have to make informed choices about the food they eat…The agency currently requires over 3,000 other ingredients, additives, and processes to be labeled; providing basic information doesn't confuse the public, it empowers them to make choices. Absent labeling, Americans are unable to choose for themselves whether to purchase GE foods…. We urge you to fully review the facts, law, and science, and side with the American public by requiring the labeling of genetically engineered foods as is done in nearly 50 countries throughout the world."
In the US there is overwhelming public demand —consistently near 95 percent— for the labeling of GE foods. The US policy of not requiring GE labeling makes it a stark outlier among developed and developing nations. Nearly 50 countries have mandatory labeling policies for GE foods including: South Korea, Japan, the United Kingdom, Brazil, China, Australia, New Zealand, the entire European Union, and many others.
In its 1992 policy statement, FDA allowed GE foods to be marketed without labeling on the basis that they were not "materially" different from other foods. However, the agency severely limited what it considered "material" by targeting only changes in food that could be recognized by taste, smell, or other senses –applying 19th century science to the regulation of 21st century food technologies.
The outdated standard has no legal basis in the statute and was adopted by FDA despite a lack of scientific studies or data to support the assumption that GE foods are not materially different from conventional foods.
Sources: Center for Food Safety Press Release, Just Label It!, and California Right to Know, beyondpesticides.org