The Food Safety Modernization Act, signed into law by President Obama on January 4, 2011, has ushered in a new era of food-safety regulation. The $1.4 billion bill—the first major reform of the nation’s food safety laws since the Great Depression—mainly expands the reach and regulatory powers of the Food and Drug Administration (FDA).
“The biggest change is the focus on prevention,” says Douglas Karas, a spokesperson for the FDA. The new legislation requires that food companies take preventive steps to avoid food contamination. They will now have to identify the critical points in their supply chain where the food they’re handling could become contaminated and then implement procedures to prevent that contamination.
The driving forces behind the Act are increased consumer awareness and globalization. High-profile outbreaks of food poisoning in recent years have shaken public confidence in the nation’s food supply. The Centers for Disease Control estimates that there are 48 million cases of food-borne illnesses in the United States each year, resulting in 128,000 hospitalizations and over 3,000 deaths. Rapid growth of imports and food ingredients from countries where safety standards are lacking has made the food supply chain more vulnerable to outbreaks of food-borne illness. Today, imports account for 15 percent of the U.S. food supply, including 80 percent of the seafood Americans consume and 50 percent to 60 percent of the fresh produce.
The nature of today’s food supply chain means contamination can turn up in more products, more quickly than in the past, causing outbreaks that affect large numbers of people. To combat this problem, the FDA will increase the frequency of inspections. High-risk domestic facilities will receive an initial inspection within the next five years and no less than every three years after that, while low-risk facilities will be visited within the next seven years. The law also significantly enhances FDA’s ability to achieve greater oversight of the millions of food products coming into the U.S. from other countries each year.
FDA experts say that the legislation, which has been supported by most of the industry trade associations, including the Food Marketing Institute (FMI) and Grocery Manufacturers of America (GMA), transforms the food safety system. The major provisions include:
- Food facilities must have a written preventive controls plan that spells out the possible problems that could affect the safety of their products. This plan would outline steps that a food facility would take to prevent or significantly minimize the likelihood of those problems occurring.
- The FDA will establish science-based standards for the safe production and harvesting of fruits and vegetables. These standards must consider not only man-made risks to fresh produce safety, but also naturally-occurring hazards—such as those posed by the soil, animals, and water in the growing area.
- The FDA is directed to increase the frequency of inspections. During the next year, the FDA must inspect at least 600 foreign food facilities and double the number of those inspections every year for the next five years. With the availability of resources, FDA will build the inspection capacity to meet these important goals.
- The FDA is authorized to mandate a recall of unsafe food if the food company fails to do it voluntarily. The law also provides a more flexible standard for administrative detention (the procedure FDA uses to keep suspect food from being moved); allows the FDA to suspend the registration of a food facility associated with unsafe food, thereby preventing it from distributing food; and directs the agency to improve its ability to track both domestic and imported foods.
With an implementation timeline in the works, food distributors are left to wonder when—and how—the new legislation will impact them.
“The way to look at this right now is a tiger is out of the cage, but he has no teeth or claws because Congress hasn’t funded the Act yet,” says P. Terrence Gaffney, an attorney with Eckert Seamans Cherin and Mello LLC, Washington, who advises on matters related to the jurisdiction of the FDA. “We have the legislation, but the FDA doesn’t have the money to implement it. And with a Republican Congress, the appropriations are going to become much more difficult than they otherwise would have.”
Rolling Out A Timeline
Funding aside, food distributors have about 18 months to develop a food safety, in they don’t already have one in place.
“Everybody in the industry knows that the law has been passed, but few have an understanding of what the law says and more importantly, what it really means for them,” says Ryan D. Shadrick Wilson, an attorney with Hogan Lovells, a law firm based in Washington.
Speaking at the 2011 Supply Chain Conference, held in Orlando, FL earlier this month by the FMI-GMA Trading Partner Alliance, Wilson said that her firm, which has been working with food industry trade associations as well as the FDA to help define the new legislation, has been flooded with calls from concerned food distributors.
“Many are unsure if the Act applies to them,” says Wilson. “There is limited exemption for very small businesses with sales less than $500,000, where the majority of food is distributed directly to consumers or restaurants, and distribution is limited to interstate or within a 275 mile radius. But if your facility is registered under Bioterrorism Act of 2002, your company has to comply. If you’re using HACCP (Hazard Analysis of Critical Control Points), you’re already meeting many of the requirements that are falling under the new Act.”
For food distributors, Wilson says the steps companies should take for daily operations include:
- Conduct hazard analysis of hazards reasonably likely to occur;
- Put into place controls designed to significantly reduce or prevent those hazards;
- Implement preventive controls through monitoring, corrective actions and verification activities;
- Verification activities include environmental and finished product testing;
- Reanalysis required every three years;
- Food safety plan and all related records available to FDA during inspection.
Wilson says that food distributors will need to put more controls in place within their operations—including sanitation for food contact surfaces and utensils, as well as food contact surfaces of equipment. “Supervisor, manager and employee training are very important,” she says. “You must also have a recall plan in place.”
Impact On Imported Goods
For food distributors handle imported goods, Geffney says there’s four things that they have to pay attention to:
- The bill requires importers to provide certification activities to ensure the imported food is safe, so the importers have to perform supplier verification activities.
- The FDA is authorized to refuse admission of imported food if the foreign facility or country refuses to allow an FDA inspector.
- The FDA is authorized to require certification based on risk criteria that the imported food is in compliance with food safety requirements.
- The bill provides an incentive to employers to take additional safety measures by the FDA to establish a voluntary program through which imports can receive if the importers have taken certain measures to ensure the food’s safety of the food.
The FDA has to create some guidelines to give those regulated by this some guidance and they haven’t finished creating all of those guidelines yet. The worst case scenario is if you get a non-compliant distributor and the FDA comes in and inspects it, the worse they would do would be to issue a 483 warning letter and it outlines where your deficiencies are and it gives you adequate time to correct those deficiencies. So no one goes to jail or gets fined.
“You can drive down one major cost—and that’s the cost of your insurance because if you can show your insurance carrier that you’ve done certain things to prevent food-born illnesses, your product liability coverage premium would likely drop.”
Retailers Impacted
In addition, retailers selling private label food products governed by the Act will be required to perform risk-based foreign supplier verification indicating that the imported food is in compliance with new FDA requirements and is not adulterated or misbranded, says Janice Lai, a partner with LeClairRyans, a law firm in Hartford, CT.
“Under the Act, private label retailers are required to provide the certification from an accredited certification entity or other assurances that their foreign-supplied food products are FDA compliant,” she says. “The Secretary can refuse entry of imported foods whose certification or assurances are not deemed valid or reliable.
“Although domestic suppliers governed by the Act have an independent duty to ensure that they are compliant with the new FDA requirements," Lai continues, “private label retailers doing business with these domestic suppliers should also make their own determination that their suppliers are indeed compliant, such as asking to see a current registration with the FDA as defined under the Act.”
"While the Act is an important step forward in further protecting America's food supply, it exempts small food producers from some regulations and oversight," says Jorge Hernandez, senior vice president of food safety and quality assurance, U.S. Foodservice, Rosemont IL. "This could raise doubts about the integrity of some channels of the food supply chain. It is therefore critical that all food producers, large and small, continue working in good faith to adhere to the highest standards of food safety throughout the harvesting, processing, distribution, storage, handling and preparation phases. We must continue to work hard each day to earn the public's trust and confidence."
U.S. Foodservice, a longstanding food safety advocate, has supported this legislation since the bill was introduced in March, 2009. The company is a founding member of the GS1 Foodservice Initiative to improve the traceability of food – from 'farm to fork'.
"As a responsible and trustworthy partner to our customers and suppliers, we view food safety as a business priority," Hernandez added. "We continuously conduct comprehensive reviews of our products and their distribution chains to identify and eliminate, or minimize to the greatest extent possible, the food safety risks along the way. We hope these new food safety regulations will encourage all levels of the food chain to create a similar food safety culture within their organizations."
What Are The Major Elements Of The Law?
The elements can be divided into five key areas:
Preventive controls: For the first time, the FDA has a legislative mandate to require comprehensive, prevention-based controls across the food supply.
Inspection and compliance: The legislation recognizes that inspection is an important means of holding industry accountable for its responsibility to produce safe food; thus, the law specifies how often FDA should inspect food producers. The FDA is committed to applying its inspection resources in a risk-based manner and adopting innovative inspection approaches.
Imported food safety: The FDA has new tools to ensure that those imported foods meet US standards and are safe for consumers. For example, for the first time, importers must verify that their foreign suppliers have adequate preventive controls in place to ensure safety, and FDA will be able to accredit qualified third party auditors to certify that foreign food facilities are complying with U.S. food safety standards.
Response: For the first time, the FDA will have mandatory recall authority for all food products. The FDA expects that it will only need to invoke this authority infrequently since the food industry largely honors its requests for voluntary recalls.
Enhanced partnerships: The legislation recognizes the importance of strengthening existing collaboration among all food safety agencies—U.S. federal, state, local, territorial, tribal and foreign—to achieve its public health goals. For example, it directs the FDA to improve training of state, local, territorial and tribal food safety officials.
Food Safety Modernization Act And Recalls
By Jeff Pepperworth, president of Inmar Reverse Logistics.
The Food Safety Modernization Act expands the reach and regulatory powers of the FDA. In summary, it adds new responsibilities for food companies, new controls on imports, enhanced enforcement powers and new fees.
With respect to recalls, the Act gives FDA mandatory recall authority, which is an expansion of its power in this area. It is important for companies to note that “Refusal or failure to comply with an order…is a prohibited act, subject to injunction and criminal prosecution.”
The responsible party is also subject to fines under the Act. In addition, grocery stores and food retailers will now be responsible for actively alerting customers of the latest product recalls and for taking specific product removal steps in their stores. The FDA will also have access to recall records for both manufacturers and retailers.
This change elevates the importance for food companies to have risk and hazard prevention measures in place, including a strong recall plan. For companies to be properly prepared in the case that the FDA mandates a recall, a recall plan should include the following:
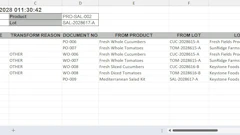
The ability to trace food and packaging throughout the supply chain;
A recall team that can begin action within 24 hours;
A plan to swiftly contact customers, food retailers, consignees and consumers;
A strategy to remove recalled product from the market;
A process to destroy or recondition recalled product according to FDA guidelines;
Flawless record keeping regarding all recall activities.